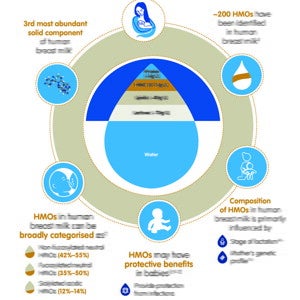
Human Milk Oligosaccharides (HMO)
Filters
- Article
- Video

Brain Development in Preterm-born Children
Dr. Nora Schneider
Restoring Gut Health in C-Section Infants: Exploring the Role of Nutrition Modulators

[Science Update] Relationship between metabolites modulated by HMOs and reduced risk of LRTIs

[Science Update] Early-life gut ecology and reduced risk for reported LRTI

[Literature library] Effects of a specific blend of 5 HMOs on gut microbiota development and maturation
[Literature library] Complexity in HMO mixture composition and gut ecosystem

[Science Update] Maternal diet and HMO profile

[Science Update] Associations between HMOs and bacterial profile and their relationships with infant body composition

[Literature library] Correlation between variations in HMO composition and intestinal epithelial barrier

[Literature library] The influence of FUT-2 and FUT-3 polymorphisms on acute respiratory infections

[Literature library] Human milk oligosaccharides and infant gut microbiota

[Literature library] Utilization of individual human milk oligosaccharides (HMOs) by Bifidobacterium strains

[Literature Library] Human milk microbiota and oligosaccharides
![[Literature library] Human milk oligosaccharide concentration throughout lactation](/sites/default/files/styles/card_m_mobile/public/2021-10/hexagons-opt.jpg?itok=QUm9__Xq)
[Literature library] Human milk oligosaccharide concentration throughout lactation

Impact of Bioactive Nutrition in Neurodevelopment - Dr. Jonas Hauser
Jonas Hauser